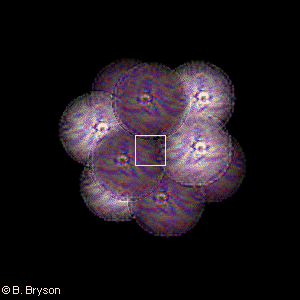
Scale: 10-14 meters = 10 fm = 10 femtometers
The Carbon NucleusHere is the stable nucleus of carbon-12, or 12C, composed of 6 protons and 6 neutrons. Carbon-12 is the standard for determining atomic weights for all the elements of the periodic table, its mass being defined as precisely 12 amu (atomic mass units). Six electrons orbit this carbon nucleus in two shells – but on this scale
there is a lot of empty space. We won't see the whole carbon
atom until we reach 10-10 – four more jumps. Although chemistry
is determined by the number of protons in the nucleus of an atom, we
are still in the realm of nuclear physics. This is the arena of stable
and unstable isotopes and the energies of fission and fusion. Copyright © 2016 by Bruce Bryson |