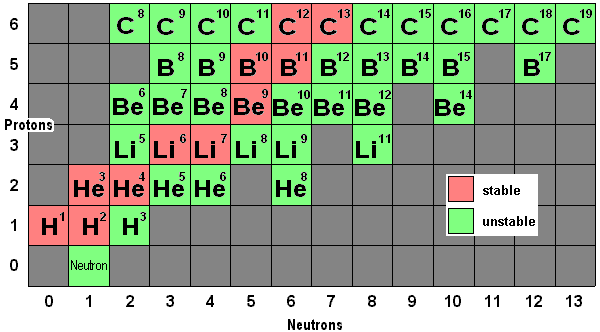
More at 10 femtometersBelow is a small section of a much larger chart that includes every known stable and unstable nuclear configuration, or isotope. The number to the upper right of the element's symbol is the total of nucleons (protons and neutrons) for that isotope. A complete chart like this one would contain a wealth of information including half life and modes of decay. It would also cover most of a wall. There are about 250 stable isotopes known and perhaps ten times as many that are unstable. Stable isotopes tend to cluster where the number of protons and neutrons are equal. For higher numbered elements, above 30 or 40, the stable isotopes begin to acquire more neutrons than protons: Uranium, element number 92, has relatively stable isotopes with 235 and 238 nucleons (92 protons and 146 neutrons for U238). Chart of Nuclear Isotopes
You can find an interactive version of a nuclear isotope chart, containing information on all known stable and unstable isotopes at Table of the Nuclides. |